Dosing &
Administering Injectafer
Replenish your patients' iron deficit with Injectafer (ferric carboxymaltose injection)
Injectafer (ferric carboxymaltose injection) is a first-line treatment for adult patients with iron deficiency anemia (IDA) and non-dialysis dependent chronic kidney disease (NDD-CKD) and adult patients with heart failure (HF) and iron deficiency (ID) and New York Heart Association (NYHA) class II/III to improve exercise capacity.1
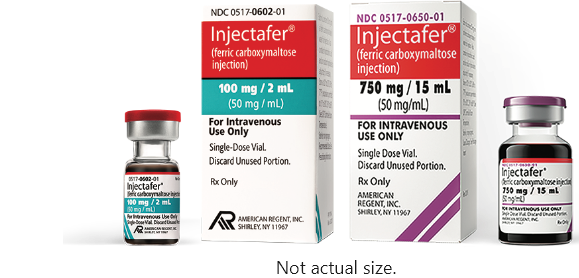
Injectafer (ferric carboxymaltose injection) provides the most iron per course of treatment—up to 1500 mg in 2 doses of 750 mg separated by at least 7 days.1-6* Injectafer is also available as a 100 mg iron/2 mL single-dose vial.1
 Artist's rendering.
Artist's rendering.
1500 mg in one course of treatment†‡


Intravenous (IV) infusion over at least 15 minutes
Slow IV push over 7.5 minutes
Recommended weight-based dosing for adult patients with ID in HF and NYHA class II/III to improve exercise capacity1
After determining your adult patients with HF and NYHA class II/III have ID, calculate the total iron needed using the dosing tables below1
| DAY 1 | |
| Hb (g/dL) | Patient body weight <70 kg or ≥70 kg |
| ≤14 | 1000 mg |
| ≥14* | 500 mg |
*There are no data available to guide Injectafer dosing in patients with Hb ≥15.
| WEEK 6† | ||||||
| Hb (g/dL) |
|
|||||
| <10 | 500 mg | 1000 mg | ||||
| ≥10* | No dose | 500 mg | ||||
*There are no data available to guide Injectafer dosing in patients with Hb ≥15.
†No week 6 dose is needed for patients with a Hb (g/dL) ≥14.
| WEEK 12, 24, AND 36‡ |
| Administer a maintenance dose of 500 mg, if serum ferritin <100 ng/mL or serum ferritin 100 ng/mL to 300 ng/mL with Transferrin Saturation (TSAT) <20% |
‡There are no data available to guide Injectafer dosing past 36 weeks.
For your adult patients with IDA who have either intolerance or an unsatisfactory response to oral iron, continue to use 2 doses of 750 mg separated by at least 7 days. For your pediatric patients 1 year of age and older with IDA use the weight based dosing in the Injectafer Prescribing Information1§II¶
A majority of patients with HF received 1500 mg or more of Injectafer (ferric carboxymaltose injection)1,7
In the Injectafer (ferric carboxymaltose injection) pivotal trials for the treatment of IDA, a fixed-dose regimen of 1500 mg was used.1 In CONFIRM-HF (a trial specific to ID treatment in certain HF patients), the mean and median total dose was 1500 mg (with a dosing range of 500 mg-3500 mg iron) to determine if intravenous Injectafer improves exercise capacity. 7
||When administered via infusion, dilute up to 750 mg of iron in no more than 250 mL of sterile 0.9% sodium chloride injection, USP, such that the concentration of the infusion is not <2 mg of iron per mL, and administer over at least 15 minutes. When administering Injectafer 500 mg or 750 mg as a slow IV push, give at the rate of approximately 100 mg (2 mL) per minute.1,8 ¶Injectafer is a first-line treatment for adult patients with IDA who have NDD-CKD.1
Injectafer (ferric carboxymaltose injection) is the most studied IV iron treatment in the world8§
Evidence for administering 1500 mg of iron per course of treatment7
In 7 clinical trials including over 4600 patients, average iron deficits were ≈1500 mg.7
Average calculated iron deficits in Injectafer (ferric carboxymaltose injection) clinical trials||
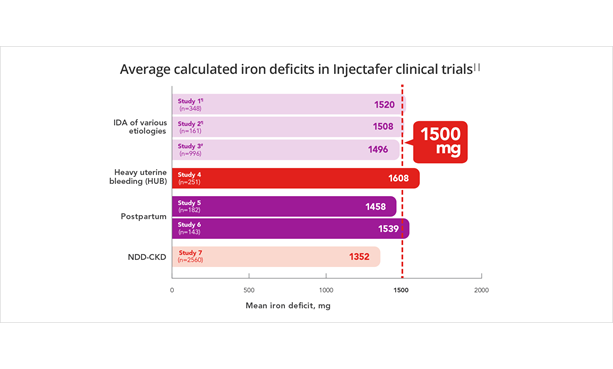
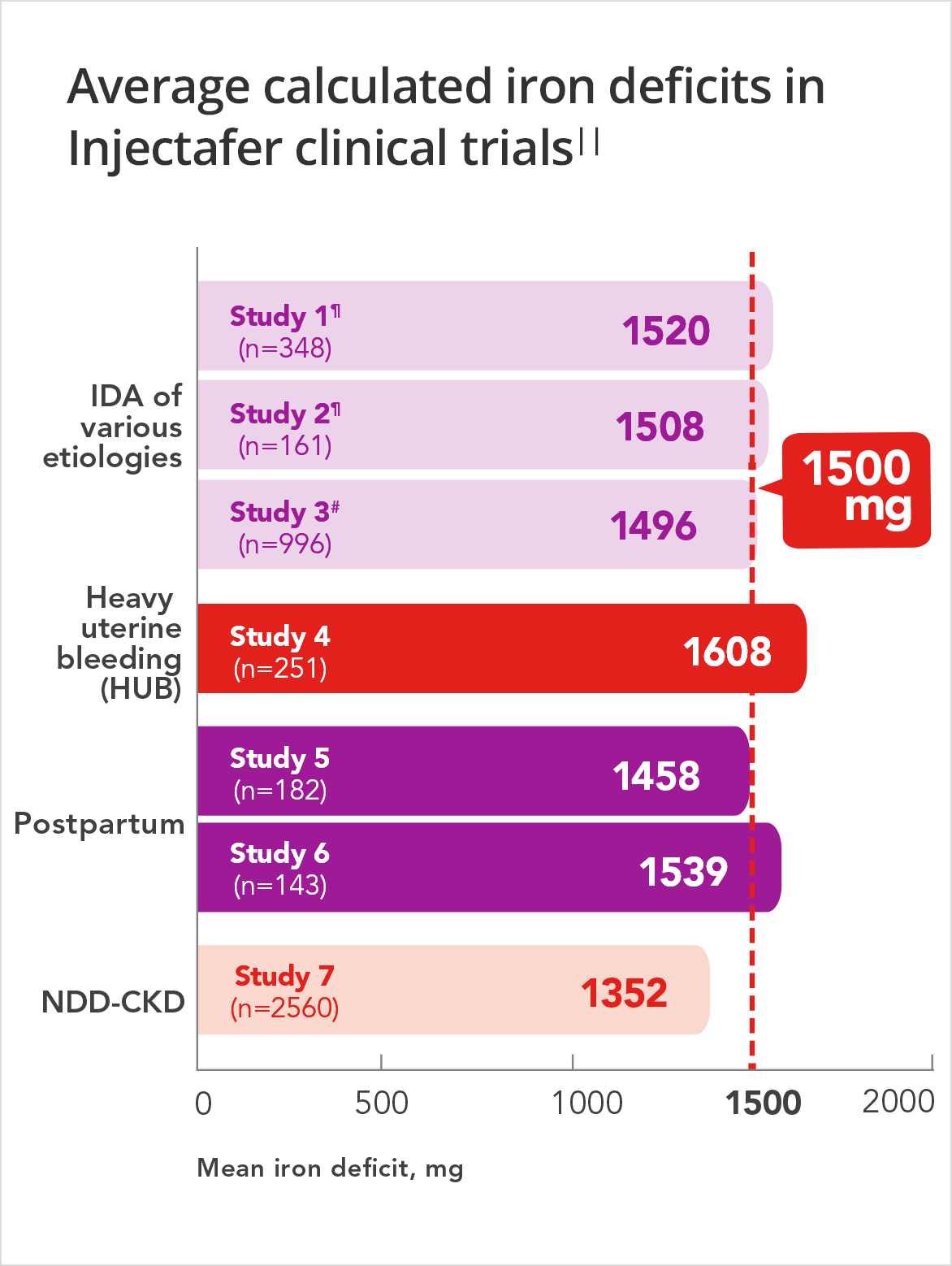
Injectafer (ferric carboxymaltose injection) is the only FDA-approved IV iron that restores up to 1500 mg of iron
in 2 administrations of 750 mg separated by at least 7 days1-6
Injectafer (ferric carboxymaltose injection) is available as a
750 mg iron/15 mL single-dose vial and as a 100 mg iron/2 mL single-dose vial.1
 Not actual size.
Not actual size.
Prior to Administration
-
Infusion bag size
Dilute Injectafer (ferric carboxymaltose injection) in up to 250 mL (but not more) of sterile 0.9% sodium chloride injection, USP, such that the concentration of the infusion is not less than 2 mg of iron per mL.1 If you need further assistance, contact American Regent at pv@americanregent.com.
-
Needle size
The infusion nurse or administering healthcare professional should assess the patient’s vein status and choose an appropriate gauge needle. If you need further assistance, contact American Regent at pv@americanregent.com.
-
Filtered tubing
Filtered tubing is not necessary. If you need further assistance, contact American Regent at pv@americanregent.com.
-
Mixing Injectafer (ferric carboxymaltose injection)
Dilute up to 750 mg of Injectafer (ferric carboxymaltose injection) in up to 250 mL (but not more) of sterile 0.9% sodium chloride injection, USP, such that the concentration of the infusion is not less than 2 mg of iron per mL. Administer over at least 15 minutes.1
At concentrations ranging from 2 mg to 4 mg of iron per mL, Injectafer (ferric carboxymaltose injection) solution is physically and chemically stable for 72 hours when stored at room temperature. To maintain stability, do not dilute to concentrations less than 2 mg iron/mL.1
Inspect parenteral drug products visually for the absence of particulate matter and discoloration prior to administration. The product contains no preservatives. Each vial of Injectafer (ferric carboxymaltose injection) is intended for single-use only. Any unused drug remaining after injection must be discarded.1
-
Storage prior to use
Store Injectafer (ferric carboxymaltose injection) at 20˚C to 25˚C (68˚F to 77˚F), excursions permitted to 15˚C to 30˚C (59˚F to 86˚F). Do not freeze.1
Administration
-
Dosing for patients with IDA who weigh 50 kg or more
Dosing for patients with IDA who weigh 50 kg (110 lb) or more: Give Injectafer (ferric carboxymaltose injection) in 2 doses separated by at least 7 days. Give each dose as 750 mg for a total cumulative dose of 1500 mg of iron per course.1
-
Dosing for patients with IDA who weigh less than 50 kg
Dosing for patients with IDA who weigh less than 50 kg (110 lb): Give Injectafer (ferric carboxymaltose injection) in 2 doses separated by at least 7 days. Give each dose as 15 mg/kg body weight.1
-
Dosing in appropriate patients with ID with HF
DAY 1 Hb (g/dL) Patient body weight
<70 kg or ≥70 kg≤14 1000 mg ≥14* 500 mg *There are no data available to guide Injectafer dosing in patients with Hb ≥15.
WEEK 6† Hb (g/dL) Patient body weight <70 kg ≥70 kg <10 500 mg 1000 mg ≥10* No dose 500 mg *There are no data available to guide Injectafer dosing in patients with Hb ≥15.
†No week 6 dose is needed for patients with a Hb (g/dL) ≥14.
WEEK 12, 24, AND 36‡ Administer a maintenance
dose of 500 mg, if serum ferritin
<100 ng/mL or serum ferritin
100 ng/mL to 300 ng/mL
with TSAT <20%‡There are no data available to guide Injectafer dosing past 36 weeks.
-
Administering by IV push
Injectafer (ferric carboxymaltose injection) may be administered as an undiluted slow IV push. Give at the rate of approximately 100 mg (2 mL) per minute.1
-
Special precautions
Injectafer (ferric carboxymaltose injection) should only be administered when personnel and therapies are immediately available for the treatment of serious hypersensitivity reactions.1
-
Occurrence of extravasation
Monitor for extravasation, and if it occurs, discontinue the Injectafer (ferric carboxymaltose injection) administration at that site immediately.1
-
Most common side effects of Injectafer (ferric carboxymaltose injection)
- The most common side effects of Injectafer (ferric carboxymaltose injection) in the pivotal trials (reported by ≥2% of study patients) were: nausea (7.2%); hypertension (4%); flushing (4%); injection site reactions (3%); erythema (3%); hypophosphatemia (2.1%); dizziness (2.1%); and vomiting (2%).
- In pivotal trials 1 and 2 for Injectafer (ferric carboxymaltose injection), serious anaphylactic/anaphylactoid reactions were reported in 0.1% (2/1775) of subjects receiving Injectafer.1
- Pivotal trials for Injectafer (ferric carboxymaltose injection) included patients with a history of oral iron intolerance, drug allergies (with the exception of known hypersensitivity to study treatments), and hypotension.11,12
Review the most common adverse events reported during pivotal trials.
References:
- Injectafer®. Package insert. American Regent, Inc.; 2025.
- Venofer® (iron sucrose) injection, USP Package insert. Shirley, NY: American Regent, Inc.; 2022.
- Ferrlecit® (sodium ferric gluconate complex in sucrose injection) Package insert. Bridgewater, NJ: sanofi-aventis US LLC; 2022.
- INFeD® (Iron Dextran Injection USP) Package insert. Parsippany, NJ: Allergan, Inc; 2021.
- Feraheme® (ferumoxytol injection) Package insert. Waltham, MA: AMAG Pharmaceuticals, Inc; 2022.
- Monoferric® (ferric derisomaltose) Package insert. Holbaek, Denmark: Pharmacosmos A/S; 2022.
- Koch TA, Myers J, Goodnough LT. Intravenous iron therapy in patients with iron deficiency anemia: dosing considerations. Anemia. 2015. doi:10.1155/2015/763576
- Data on File. American Regent. Shirley, NY.
- Hussain I, Bhoyroo J, Butcher A, Koch TA, He A, Bregman DB. Direct comparison of the safety and efficacy of ferric carboxymaltose versus iron dextran in patients with iron deficiency anemia. Anemia. 2013. doi:10.1155/2013/169107
- Barish CF, Koch T, Butcher A, Morris D, Bregman DB. Safety and efficacy of intravenous ferric carboxymaltose (750 mg) in the treatment of iron deficiency anemia: two randomized, controlled trials. Anemia. 2012. doi:10.1155/2012/172104
- Onken JE, Bregman OB, Harrington RA, et aI. A multicenter, randomized, active-controlled study to investigate the efficacy and safety of intravenous ferric carboxymaltose in patients with iron deficiency anemia. Transfusion. 2014;54(2):306-315
- Onken JE, Bregman DB, Harrington RA, et al. Ferric carboxymaltose in patients with iron-deficiency anemia and impaired renal function: the REPAIR-IDA trial. Nephrol Dial Transplant. 2014;29(4):833-842.
-
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Injectafer is contraindicated in patients with hypersensitivity to Injectafer or any of its inactive components.
WARNINGS AND PRECAUTIONS
Symptomatic Hypophosphatemia
Symptomatic hypophosphatemia with serious outcomes including osteomalacia and fractures requiring clinical intervention has been reported in patients treated with Injectafer in the post-marketing setting. These cases have occurred mostly after repeated exposure to Injectafer in patients with no reported history of renal impairment. However, symptomatic hypophosphatemia has been reported after one dose. Possible risk factors for hypophosphatemia include a history of gastrointestinal disorders associated with malabsorption of fat-soluble vitamins or phosphate, inflammatory bowel disease, concurrent or prior use of medications that affect proximal renal tubular function, hyperparathyroidism, vitamin D deficiency, malnutrition, and hereditary hemorrhagic telangiectasia (HHT or Osler-Weber-Rendu syndrome). In most cases, hypophosphatemia resolved within three months.
Correct pre-existing hypophosphatemia prior to initiating therapy with Injectafer. Monitor serum phosphate levels in patients at risk for chronic low serum phosphate. Check serum phosphate levels prior to a repeat course of treatment in patients at risk for low serum phosphate and in any patient who receives a second course of therapy within three months. Treat hypophosphatemia as medically indicated.
Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylactic-type reactions, some of which have been life-threatening and fatal, have been reported in patients receiving Injectafer. Patients may present with shock, clinically significant hypotension, loss of consciousness, and/or collapse. Monitor patients for signs and symptoms of hypersensitivity during and after Injectafer administration for at least 30 minutes and until clinically stable following completion of the infusion. Only administer Injectafer when personnel and therapies are immediately available for the treatment of serious hypersensitivity reactions. In clinical trials, serious anaphylactic/anaphylactoid reactions were reported in 0.1% (2/1775) of subjects receiving Injectafer. Other serious or severe adverse reactions potentially associated with hypersensitivity which included, but were not limited to, pruritus, rash, urticaria, wheezing, or hypotension were reported in 1.5% (26/1775) of these subjects.
Hypertension
In clinical studies, hypertension was reported in 4% (67/1775) of subjects in clinical trials 1 and 2. Transient elevations in systolic blood pressure, sometimes occurring with facial flushing, dizziness, or nausea were observed in 6% (106/1775) of subjects in these two clinical trials. These elevations generally occurred immediately after dosing and resolved within 30 minutes. Monitor patients for signs and symptoms of hypertension following each Injectafer administration.
Laboratory Test Alterations
In the 24 hours following administration of Injectafer, laboratory assays may overestimate serum iron and transferrin bound iron by also measuring the iron in Injectafer.
ADVERSE REACTIONS
Adults
In two randomized clinical studies [Studies 1 and 2], a total of 1775 patients were exposed to Injectafer, 15 mg/kg of body weight, up to a maximum single dose of 750 mg of iron on two occasions, separated by at least 7 days, up to a cumulative dose of 1500 mg of iron. Adverse reactions reported by >2% of Injectafer-treated patients were nausea (7.2%); hypertension (4%); flushing (4%); injection site reactions (3%); erythema (3%); hypophosphatemia (2.1%); and dizziness (2.1%).
Pediatric
The safety of Injectafer in pediatric patients was evaluated in Study 3. Study 3 was a randomized, active-controlled study in which 40 patients (1 to 12 years of age: 10 patients, 12 to 17 years of age: 30 patients) received Injectafer 15 mg/kg to a maximum single dose of 750 mg (whichever was smaller) on Days 0 and 7 for a maximum total dose of 1500 mg; 38 patients evaluable for safety in the control arm received an age-dependent formulation of oral ferrous sulfate for 28 days. The median age of patients who received Injectafer was 14.5 years (range, 1-17); 83% were female; 88% White and 13% Black. The most common adverse reactions (≥4%) were hypophosphatemia (13%), injection site reactions (8%), rash (8%), headache (5%), and vomiting (5%).
Patients with Iron Deficiency and Heart Failure
The safety of Injectafer was evaluated in adult patients with iron deficiency and heart failure in randomized controlled trials FAIR-HF (NCT00520780), CONFIRM-HF (NCT01453608) and AFFIRM-AHF (NCT02937454) in which 1016 patients received Injectafer versus 857 received placebo. The overall safety profile of Injectafer was consistent across the studied indications.
Post-Marketing Experience
The following adverse reactions have been identified during post approval use of Injectafer. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been reported from the post-marketing spontaneous reports with Injectafer: cardiac disorders: tachycardia; general disorders and administration site conditions: chest discomfort, chills, pyrexia; metabolism and nutrition disorders: hypophosphatemia; musculoskeletal and connective tissue disorders: arthralgia, back pain, hypophosphatemic osteomalacia; nervous system disorders: syncope; respiratory, thoracic and mediastinal disorders: dyspnea; skin and subcutaneous tissue disorders: angioedema, erythema, pruritus, urticaria; pregnancy: fetal bradycardia.
USE IN SPECIFIC POPULATIONS
Pregnancy – Fetal/Neonatal Adverse Reactions
Severe adverse reactions including circulatory failure (severe hypotension, shock including in the context of anaphylactic reaction) may occur in pregnant women with parenteral iron products (such as Injectafer) which may cause fetal bradycardia, especially during the second and third trimester.
INDICATIONS
Injectafer® (ferric carboxymaltose injection) is indicated for the treatment of iron deficiency anemia (IDA) in adult and pediatric patients 1 year of age and older who have either intolerance or an unsatisfactory response to oral iron, and in adult patients who have non-dialysis dependent chronic kidney disease. Injectafer is also indicated for iron deficiency in adult patients with heart failure and New York Heart Association class II/III to improve exercise capacity.
You are encouraged to report Adverse Drug Events to American Regent, Inc. at 1-800-734-9236 or to the FDA by visiting www.fda.gov/medwatch or calling 1-800-FDA-1088.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
Injectafer is contraindicated in patients with hypersensitivity to Injectafer or any of its inactive components.
WARNINGS AND PRECAUTIONS
Symptomatic Hypophosphatemia
Symptomatic hypophosphatemia with serious outcomes including osteomalacia and fractures requiring clinical intervention has been reported in patients treated with Injectafer in the post-marketing setting. These cases have occurred mostly after repeated exposure to Injectafer in patients with no reported history of renal impairment. However, symptomatic hypophosphatemia has been reported after one dose. Possible risk factors for hypophosphatemia include a history of gastrointestinal disorders associated with malabsorption of fat-soluble vitamins or phosphate, inflammatory bowel disease, concurrent or prior use of medications that affect proximal renal tubular function, hyperparathyroidism, vitamin D deficiency, malnutrition, and hereditary hemorrhagic telangiectasia (HHT or Osler-Weber-Rendu syndrome). In most cases, hypophosphatemia resolved within three months.
Correct pre-existing hypophosphatemia prior to initiating therapy with Injectafer. Monitor serum phosphate levels in patients at risk for chronic low serum phosphate. Check serum phosphate levels prior to a repeat course of treatment in patients at risk for low serum phosphate and in any patient who receives a second course of therapy within three months. Treat hypophosphatemia as medically indicated.
Hypersensitivity Reactions
Serious hypersensitivity reactions, including anaphylactic-type reactions, some of which have been life-threatening and fatal, have been reported in patients receiving Injectafer. Patients may present with shock, clinically significant hypotension, loss of consciousness, and/or collapse. Monitor patients for signs and symptoms of hypersensitivity during and after Injectafer administration for at least 30 minutes and until clinically stable following completion of the infusion. Only administer Injectafer when personnel and therapies are immediately available for the treatment of serious hypersensitivity reactions. In clinical trials, serious anaphylactic/anaphylactoid reactions were reported in 0.1% (2/1775) of subjects receiving Injectafer. Other serious or severe adverse reactions potentially associated with hypersensitivity which included, but were not limited to, pruritus, rash, urticaria, wheezing, or hypotension were reported in 1.5% (26/1775) of these subjects.
Hypertension
In clinical studies, hypertension was reported in 4% (67/1775) of subjects in clinical trials 1 and 2. Transient elevations in systolic blood pressure, sometimes occurring with facial flushing, dizziness, or nausea were observed in 6% (106/1775) of subjects in these two clinical trials. These elevations generally occurred immediately after dosing and resolved within 30 minutes. Monitor patients for signs and symptoms of hypertension following each Injectafer administration.
Laboratory Test Alterations
In the 24 hours following administration of Injectafer, laboratory assays may overestimate serum iron and transferrin bound iron by also measuring the iron in Injectafer.
ADVERSE REACTIONS
Adults
In two randomized clinical studies [Studies 1 and 2], a total of 1775 patients were exposed to Injectafer, 15 mg/kg of body weight, up to a maximum single dose of 750 mg of iron on two occasions, separated by at least 7 days, up to a cumulative dose of 1500 mg of iron. Adverse reactions reported by >2% of Injectafer-treated patients were nausea (7.2%); hypertension (4%); flushing (4%); injection site reactions (3%); erythema (3%); hypophosphatemia (2.1%); and dizziness (2.1%).
Pediatric
The safety of Injectafer in pediatric patients was evaluated in Study 3. Study 3 was a randomized, active-controlled study in which 40 patients (1 to 12 years of age: 10 patients, 12 to 17 years of age: 30 patients) received Injectafer 15 mg/kg to a maximum single dose of 750 mg (whichever was smaller) on Days 0 and 7 for a maximum total dose of 1500 mg; 38 patients evaluable for safety in the control arm received an age-dependent formulation of oral ferrous sulfate for 28 days. The median age of patients who received Injectafer was 14.5 years (range, 1-17); 83% were female; 88% White and 13% Black. The most common adverse reactions (≥4%) were hypophosphatemia (13%), injection site reactions (8%), rash (8%), headache (5%), and vomiting (5%).
Patients with Iron Deficiency and Heart Failure
The safety of Injectafer was evaluated in adult patients with iron deficiency and heart failure in randomized controlled trials FAIR-HF (NCT00520780), CONFIRM-HF (NCT01453608) and AFFIRM-AHF (NCT02937454) in which 1016 patients received Injectafer versus 857 received placebo. The overall safety profile of Injectafer was consistent across the studied indications.
Post-Marketing Experience
The following adverse reactions have been identified during post approval use of Injectafer. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
The following adverse reactions have been reported from the post-marketing spontaneous reports with Injectafer: cardiac disorders: tachycardia; general disorders and administration site conditions: chest discomfort, chills, pyrexia; metabolism and nutrition disorders: hypophosphatemia; musculoskeletal and connective tissue disorders: arthralgia, back pain, hypophosphatemic osteomalacia; nervous system disorders: syncope; respiratory, thoracic and mediastinal disorders: dyspnea; skin and subcutaneous tissue disorders: angioedema, erythema, pruritus, urticaria; pregnancy: fetal bradycardia.
USE IN SPECIFIC POPULATIONS
Pregnancy – Fetal/Neonatal Adverse Reactions
Severe adverse reactions including circulatory failure (severe hypotension, shock including in the context of anaphylactic reaction) may occur in pregnant women with parenteral iron products (such as Injectafer) which may cause fetal bradycardia, especially during the second and third trimester.
INDICATIONS
Injectafer® (ferric carboxymaltose injection) is indicated for the treatment of iron deficiency anemia (IDA) in adult and pediatric patients 1 year of age and older who have either intolerance or an unsatisfactory response to oral iron, and in adult patients who have non-dialysis dependent chronic kidney disease. Injectafer is also indicated for iron deficiency in adult patients with heart failure and New York Heart Association class II/III to improve exercise capacity.
You are encouraged to report Adverse Drug Events to American Regent, Inc. at 1-800-734-9236 or to the FDA by visiting www.fda.gov/medwatch or calling 1-800-FDA-1088.




